1 Therapeutic Products Act. 15 December 2000 (Status as of 1 January 2019), The Federal Assembly of the Swiss Confederation:
https://www.admin.ch/opc/en/classified-compilation/20002716/index.html.
2 Regulation (EC) No 1901/2006 on medicinal products for paediatric use. 2006, European Parliament and Council of the European Union:
https://eur-lex.europa.eu/legal-content/DE/TXT/PDF/uri=CELEX:32006R1901&from=EN.
3 Wimmer S, Rascher W, and Neubert A, How often do SmPCs Contain Contraindications and Special Warnings that are Specific for the Paediatric Population. Klin Padiatr, 2019. 231(4): p. 191–198.
4 Haniman A, et al. Analyse de la situation concernant les mesures liées aux autorisations de mise sur le marché de médicaments administrés en pédiatrie: «plan d’investigation pédiatrique (PIP)» et «médicaments importants contre des maladies rares». 2022, Interface Politikstudien Forschung Bratung AG:
https://www.bag.admin.ch/bag/fr/home/das-bag/publikationen/evaluationsberichte/evalber-biomedizin-forschung.html
5 Tilen R, et al. Information in Swiss drug labels about use in children is limited and need standardisation. 2022: GSASA Kongress Luzern.
6 ASSM, Distinction entre thérapie standard et thérapie expérimentale dans le cadre individuel (2014, adapté en 2015).
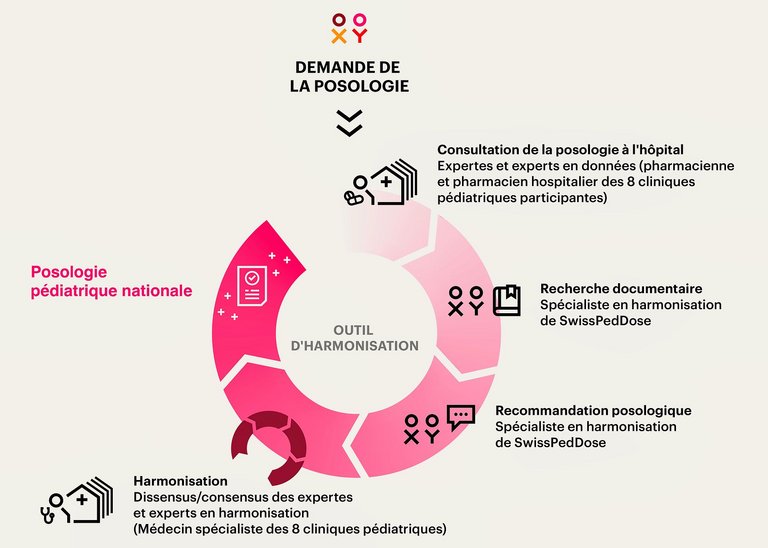
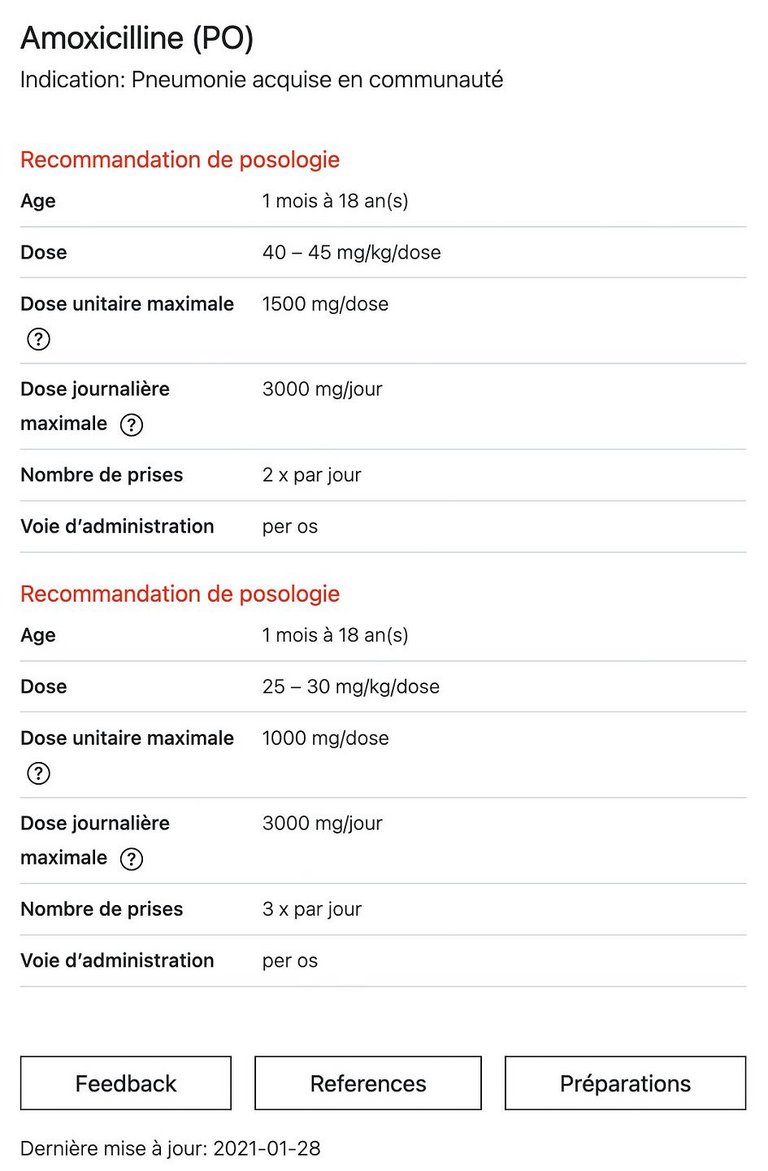
7 Tilen R, et al. Development of the Swiss Database for dosing medicinal products in pediatrics. Eur J Pediatr, 2022. 181(3): p. 1221-1231.
8 Gotta V, et al. In silico studies to evaluate dosing intervals associated with low risk of amikacin accumulation in preterm neonates. 2022: GSASA Kongress Luzern.
9 Ravix A, et al. Dosing optimization of amoxicillin in children treated for Lyme disease. 2022: GSASA Kongress Luzern.
10 Paioni P, et al. Gentamicin Population Pharmacokinetics in Pediatric Patients-A Prospective Study with Data Analysis Using the saemix Package in R. Pharmaceutics, 2021. 13: 1596.
11 Tilen R., et al. Pharmacogenetic Analysis of Voriconazole Treatment in Children. Pharmaceutics, 2022. 14:1289.
12 Kägi W, et al. Analyse de la situation concernant la mise en place de la base de données nationale de dosages de médicaments administrés en pédiatrie «SwissPedDose». 2022, BSS Volkswirtschaftliche Beratung:
https://www.bag.admin.ch/bag/fr/home/das-bag/publikationen/evaluationsberichte/evalber-biomedizin-forschung.html.